 Like almost all polymers, protein fold and twist, forming into the secondary structure, which is rigidified by hydrogen bonding between the carbonyl oxygens and amide hydrogens in the backbone, i.e. The sequence of the amino acids in the polypeptide backbone is known as the primary structure of the protein. Proteins are characterized by amide linkages (-N(H)-C(O)-) formed by the condensation of amino acids. Major families of biopolymers are polysaccharides (carbohydrates), peptides, and polynucleotides. Some uncommon but illustrative inorganic polymers include polythiazyl ((SN)x) with alternating S and N atoms, and polyphosphates ((PO 3 −) n). The silicon atoms bear two substituents, usually methyl as in the case of polydimethylsiloxane. Their backbond is composed of alternating silicon and oxygen atoms, i.e. Siloxanes are a premier example of an inorganic polymer, even though they have extensive organic substituents. Inorganic polymers Polydimethylsiloxane is classified as an " inorganic polymer", because the backbone lacks carbon. Major commercial products are polyethyleneterephthalate ("PET"), ((C 6H 4CO 2C 2H 4OC(O)) n) and nylon-6 ((NH(CH 2) 5C(O)) n). They have respectively -C(O)-O- and -C(O)-NH- groups in their backbones in addition to chains of carbon. Other major classes of organic polymers are polyesters and polyamides. Examples include polyolefins such as polyethylene ((CH 2CH 2) n) and many substituted derivative ((CH 2CH(R)) n) such as polystyrene (R = C 6H 5), polypropylene (R = CH 3), and acrylates (R = CO 2R'). Organic polymers Formation of polystyrene, a polymer with an organic backbone.Ĭommon synthetic polymers have main chains composed of carbon, i.e. Crystallization in its turn affects the optical properties of the polymers, its optical band gap and electronic levels. polythiophenes) in thin films and in solution. The polymers with rigid backbones are prone to crystallization (e.g. For example, in polysiloxanes (silicone), the backbone chain is very flexible, which results in a very low glass transition temperature of −123 ☌ (−189 ☏ 150 K). its flexibility, determines the properties of the polymer (such as the glass transition temperature). Polymers are often classified according to the elements in the main chains. In polymer science, the polymer chain or simply backbone of a polymer is the main chain of a polymer. Selected which leads to the simplest representation of the It is extremely important to understand the physical forces behind a peptide bond, as this allows scientists to design accurate, predictive models of three-dimensional protein structures.That linear chain to which all other chains, long or short or both,Ĭould equally be considered to be the main chain, that one is A mistake in the translation process can lead to protein mis-folding, and in turn, disease. Proteins can be as small as forty-four amino acids, or as large as thirty-five thousand. 
Peptide bonds are made within ribosomes during a process called «translation» to form polypeptides, which then undergo various molecular processing and modification, before folding into a three-dimensional shape, which we call a protein. 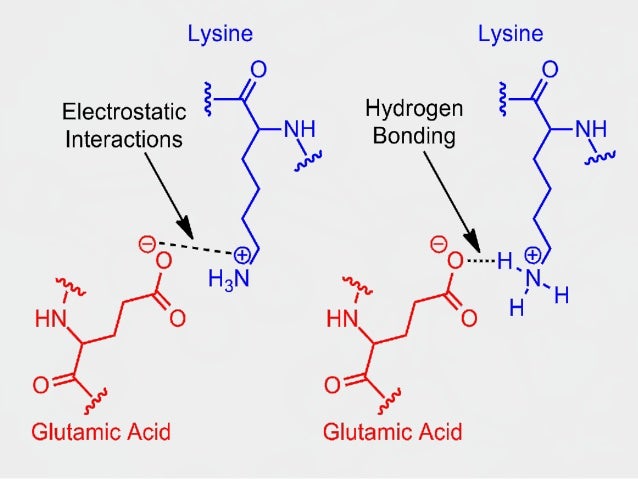
For instance, there is currently much interest in antibody-drug conjugates these pair fragmented antibodies with pharmacologically active compounds in order to specifically target cancer tumors, among other things. The ability to predictably split peptide bonds is vital to a number of different fields of study. Reversing a peptide bond without an enzyme is extremely difficult, thus this process is usually mediated by an enzyme called a protease, such as subtilisin, which is frequently added to laundry detergent to cleave many protein contaminants. It may therefore be counterintuitive to learn that peptide bonds are quite stable kinetically: the lifetime of a peptide bond in aqueous solution is approximately 1000 years. Hence, the biosynthesis of a peptide bond requires an input of free energy. One interesting thing to note is that the equilibrium of this reaction lies on the side of hydrolysis rather than synthesis. For most peptides the cis-form is about 1000 times less stable than the trans-form. In such cases, the cis form is more stable than usual since the proline side-chain offers less of a hindrance. However, cis forms can occur in peptide bonds that precede a proline residue. In naturally occurring peptides most peptide bonds are in the trans configuration. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
